Description
Meridian Bioscience Alethia® Pertussis External Controls
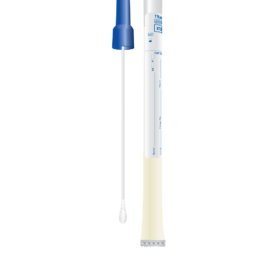
Molecular assay for the detection of Bordetella pertussis. Alethia® provides a quick, accurate and reliable molecular assay for Bordetella pertussis detection.
The CDC, IDSA and AAP recognize molecular testing as an important tool to diagnose B. pertussis1,2 due to improved sensitivity and rapid turnaround time.
Traditional culture requires a 7-10-day turnaround, has a variable sensitivity (12-60%) and requires subjective analysis. Send-out costs and delayed results are a burden on the laboratory.
- In recent years, there have been more reported cases of Bordetella pertussis (Whooping Cough) in the U.S. than any other time since 1955
- Studies indicate vaccinations for Bordetella pertussis don’t always provide lifelong protection.
- Alethia Pertussis has a simple procedure with low invalid rates that eliminate the need for repeat testing.
Is your current testing method accurate?
- How do missed positives affect patient treatment?
- How long does it take your lab to get results with your current method?
Results that Matter
- With molecular results in less than one hour, long turnaround times and send out costs can be eliminated.
Improve Outcomes for Patients
- Bordetella pertussis illness can be clinically indistinguishable from other respiratory infections.
- Targeted detection can help physicians treat patients appropriately.
- The molecular accuracy of Alethia Pertussis provides a clear and objective evaluation of positive/ negative test results. This can help to reduce the risk of spreading illness to others and allow for appropriate treatment as soon as possible.